The following data were collected for the reaction of ICl and H_2 at a certain temperature.
2ICl + H_2 → I_2 + 2HCl
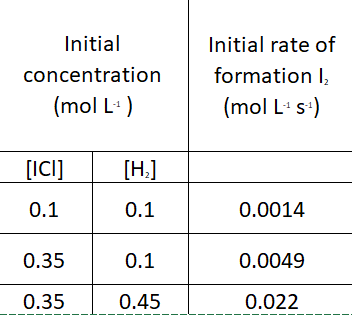
The rate law for the reaction is ……
a. Rate = k [ICl]
b. Rate = k [ICl] [H_2]
c. Rate = k [ICl]^2 [H_2]
d. Rate = k [ICl] [H_2]^2
e. Rate = k [ICl]^2 [H_2]^2